Acute respiratory illness (ARI) includes a range of symptoms like cough, fever, runny nose and sore throat. Acute respiratory illnesses are often caused by viral infections affecting the airways including influenza, SARS-CoV-2 and common cold viruses.
Surveillance of acute respiratory illness allows for tracking of influenza (flu), as well as other viruses that cause similar symptoms, such as SARS-CoV-2, respiratory syncytial virus (RSV), and those that could be newly emerging. Respiratory virus surveillance systems are in place to monitor virus activity, detect epidemics or pandemics, inform vaccination policy and vaccine strain selection, and guide public health control measures in New Zealand and globally.
Information on influenza and other respiratory viruses is presented weekly on a dashboard throughout the winter surveillance period (May to October) and as required outside of the winter period. See the acute respiratory illness dashboard and annual reports.
For any questions related to surveillance, please contact surv.queries@esr.cri.nz
-
Surveillance objectives
The overarching goal of respiratory virus surveillance is to minimise the impact of disease by providing useful information to public health authorities and communities so they may better plan for appropriate health promotion, disease protection and control measures.
The specific aims of acute respiratory illness surveillance are to:
- To actively monitor and better understand the patterns of activity (e.g. seasonality, severity) of influenza, SARS-CoV- 2 and other respiratory viruses;
- Describe the burden of ILI- and SARI-associated influenza, SARS-CoV-2 and other respiratory viruses in New Zealand, including among priority populations;
- Describe characteristics of locally circulating influenza, SARS-CoV-2 and other respiratory viruses;
- Inform influenza and COVID-19 vaccination and treatment decisions to reduce the burden of disease and support equity of outcomes;
- Facilitate decision-making for pandemic influenza risk management nationally and globally.
-
Overview of respiratory virus surveillance
The burden of respiratory viruses, including influenza, RSV and SARS-CoV-2 can vary widely throughout and between years. While the effects of these viruses vary, it places a substantial burden on the people’s health and the health system every year.
ARI surveillance systems collect information which together builds a picture of the burden of disease from influenza and other acute respiratory viruses in New Zealand and allows us to assess how well important public health protective measures, such as vaccination, are working. These surveillance systems operate in the community, primary care, hospitals and laboratories to capture disease presentations at different levels of severity (Figure 1). These are described in detail in surveillance systems.
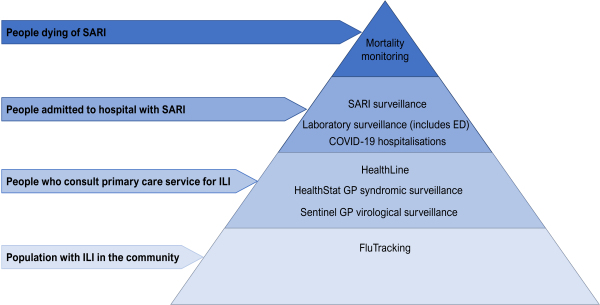
Figure 1: Burden of respiratory illness pyramid and surveillance systems employed at varying levels of severity
-
Community and hospital surveillance systems
Case definitions
Influenza-like illness (ILI): ILI is defined by the World Health Organisation (WHO) as an acute respiratory infection with a history of fever (or measured fever of ≥38°C) AND cough.
Severe acute respiratory infection (SARI): SARI is defined by the WHO as the acute onset of cough and fever in the previous 10 days requiring hospitalisation.
- HealthStat sentinel general practice surveillance of influenza-like illness consultations – This system monitors the number of people who have presented to their general practice with influenza-like illness in the past week. Consultation information is extracted from around 300 general practice clinics nationally. Get more information on HealthStat.
- HealthLine – This system monitors the number of calls made to Healthline (the free, national, 24-hour telephone health service) for acute respiratory illness advice each week. Calls are triaged using electronic clinical decision support software. Those coded as cold/flu, cough, croup, fever, general aches, headache and/or sore throat are counted as ILI. Non-symptomatic calls (30% of HealthLine calls) are excluded.
- FluTracking – This system relies on voluntary participation by the general public to self-report illness online. It was launched in New Zealand in 2018 and is used to monitor community-level ILI trends. In 2022, there were over 64,700 people who completed at least one FluTracking survey and an average of 37,200 responses were received per week. Participants are given the option to opt out outside of the influenza season (May to October), accordingly the sample size will be smaller at this time. Those who record experiencing a fever and cough are counted as ILI. See more information on FluTracking here. More detailed analysis of FluTracking data, and historical reports are also available.
- Sentinel GP virological surveillance - ESR works with a network of 50-100 sentinel general practice clinics around the country who take a respiratory swab from a subset of patients presenting to their clinic each week with ILI. These swabs are tested for a range of respiratory viruses at the ESR laboratory (see viral identification and characterisation methods). This system provides information on the different viruses that are circulating and causing illness in the community.
- Sentinel hospital surveillance – This system monitors patients admitted to hospital overnight with SARI in the four hospitals of the Auckland and Counties Manukau districts. Research nurses collect information on SARI patients in general wards and intensive care units (ICUs). Nasopharyngeal swabs are offered to patients if not already taken as part of clinical care and are tested for influenza and other respiratory viruses. Which viruses are tested for varies and is dependent on several factors, including clinical decision making, laboratory capacity and patient characteristics. The system operates from April to October in general wards and year-round in intensive care units (ICUs), though the surveillance period has been extended since the COVID-19 pandemic begun.
- Non-sentinel laboratory surveillance – A voluntary network of hospital laboratories provide data on detections of respiratory viruses in samples taken from individuals tested in hospital, emergency departments and in the community. Results should be interpreted with caution as these are from samples taken for various reasons as part of clinical care and are not systematically collected for surveillance. Samples undergoing testing and the viruses being tested for depend on several factors, including clinical decision making, laboratory capacity and patient demographic, and results will vary by region and over time without necessarily reflecting true epidemiological trends. In addition, negative results are not currently supplied to ESR, and so test positivity is not known.
- COVID-19 notifiable disease surveillance - COVID-19 became a notifiable disease in New Zealand on 30 January 2020. The Ministry of Health collects data on reported cases, hospitalisations and deaths. This information is reported on the COVID-19 Trends and Insights Dashboard.
ESR undertakes testing of wastewater throughout New Zealand on behalf of the Ministry for Health for the presence of SARS-CoV-2. The ESR wastewater dashboard can be found here.
ESR and collaborators also conduct genomic analysis of SARS-CoV-2 samples from COVID-19 cases and wastewater, this is reported in the COVID-19 Genomics Insights Dashboard.
-
Viral identification and characterisation methods
Swabs collected through sentinel GP ILI surveillance are tested at ESR for influenza and other respiratory viruses (respiratory syncytial virus (RSV), parainfluenza virus 1–3, human metapneumovirus, rhinovirus and adenovirus). Swabs collected through sentinel hospital surveillance are tested at hospital labs for influenza and other viruses. Results from these tests for SARI patients are forwarded to ESR for integration into surveillance.
Samples forwarded to ESR that are found to be influenza-positive undergo antigenic, genetic and antiviral characterization either at ESR or the WHO collaborating centres in Melbourne.
-
Analytic Methods
Rate Calculations
Population data used to calculate rates of hospitalisations and HealthLine calls are derived from mid-year population estimates published by Statistics New Zealand. HealthStat general practice visit rates are calculated using registered patient populations. Registered patient counts are limited further by participating clinicians, if not all practice clinicians are contributing to surveillance.
Presented rates are unadjusted.
MEM Thresholds
MEM is a standardised method of reporting influenza activity adopted by the European Centre for Disease Prevention and Control that allows intra- and inter- country comparisons. MEM defines the baseline influenza activity in historical data and establishes an epidemic threshold above which the weekly rates are considered to be in the epidemic period. Based on the historical data, influenza activity intensity is then also described according to categories as follows: (i) baseline: weekly rate is below or at the baseline epidemic threshold; (ii) low: weekly rate is above the baseline threshold and below the low average seasonal activity threshold; (iii) medium: weekly rate is between the low and medium activity threshold (iv) high: weekly rate is between the medium and high activity thresholds; (v) very high: weekly rate is above the high activity threshold.
The historical data used to calculate the MEM thresholds for the GP ILI surveillance graph has been collected by ESR over the years 2000 to 2017, excluding the pandemic year 2009. The thresholds are set at the 40%, 90%, and 97.5% confidence intervals, and labelled "Low seasonal level", "Moderate Seasonal Level", and "High Seasonal Level". The level at which the season is defined as having started is labelled "Baseline Seasonal Level".
